
Technology
Overview
BioMendics is evaluating its patented drug platform of small-molecule liquid crystals. Currently, BioMendics is focused on unmet medical needs in rare genetic skin diseases such as epidermolysis bullosa (EB), a serious and life-threatening condition with no current treatment options. These small molecules promote keratin intermediate filament (IF) network stabilization, tissue regeneration, are anti-inflammatory, antibacterial, and antiviral.
TolaSure
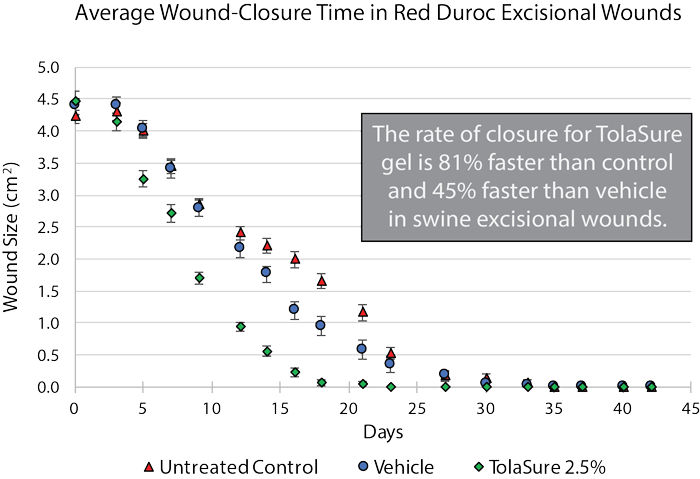
BioMendics lead topical formulation TolaSure is a patented topical gel formulation containing small-molecule liquid crystal. TolaSure has been shown to cut wound healing times in half while being non-immunogenic and anti-inflammatory to the skin. Multiple studies in full thickness wounds treated with TolaSure showed healing rates from 45-75% faster than moist wound care-treated controls in multiple species including: mice, rats, and various swine models. BioMendics is currently completing a Phase I clinical trial of TolaSure in acute punch biopsy wounds created in healthy volunteers. To date there have been no adverse events attributed to the investigational drug product.
Pipeline for TolaSure
Optimization
Epidermolysis Bullosa
The incidence rate of EB is between 42-70,000 patients or 1 in 20,000 births in the United States with estimates as high as 500,000 individuals worldwide. There are currently no known or approved therapeutic interventions for EB.
EB refers to a group of rare, genetic, connective tissue disorders that affects the mechanical integrity of the epithelial layers of the skin. The condition manifests as delamination of the skin initiating a chronic cycle of severe blistering, chronic wounds, pain, inflammation, itch, secondary infections, mobility losses, and decreased life expectancy.
Epidermolysis bullosa simplex (EBS) is the most commonly occurring EB type with three variants based on the most recent classification system: localized (previously Weber-Cockayne), intermediate (previously Köbner), and severe (previously Dowling-Meara). Severe EBS is characterized by extremely fragile skin which manifests in more frequent and more severe blistering, a herpetiform blistering pattern, and an increased risk of life-threatening complications within the first year of life. Resulting blister formation occurs at the dermo-epidermal junction as these mutations result in a mechanically compromised intermediate filament (IF) network in the basement membrane of the epidermis. While this network may appear normal, upon stressing (e.g., mechanical trauma or elevated temperature) the keratin IF network collapses into fragments and assembles into aggregates (collapsed tonofilaments) that do not confer mechanical resilience. Preclinical testing revealed a more stabilized keratin IF network after treatment with TolaSure. As a result of these preclinical studies in conjunction with a strong safety profile for our Phase I and II clinical studies in acute wounds, BioMendics is currently recruiting for a Phase I/IIa clinical trial for epidermolysis bullosa simplex (EBS) patients.
Wound Healing
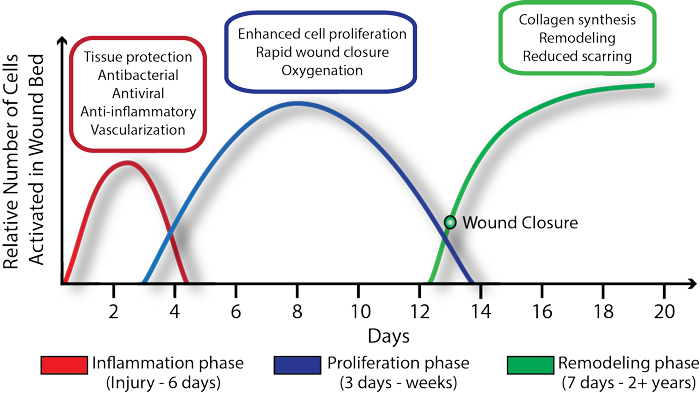
Unlike traditional wound care products that target a specific phase of the healing process TolaSure targets keystone signaling pathways that interact at each stage of the wound healing process from inflammation to remodeling. The faster healing rates seen in preclinical and clinical studies are attributed to this multipronged approach of reduction of inflammation, increase in cell proliferation, reduced scarring and protection from bacterial infection.
Inflammatory Skin Conditions
TolaSure’s ability to suppress inflammation is key for inflammatory skin conditions such as psoriasis, dermatitis, and scleroderma. BioMendics is currently in the preclinical stages of developing formulations to combat these conditions.
Antibacterial
According the CDC antibiotic resistance is a global threat, and the development of new antibiotics is critical to the looming post-antibiotic era. TolaSure has shown promising preclinical results against a variety of highly resistant bacterial strains including: MRSA, E. Coli, P. aeruginosa, and A. baumannii. TolaSure is also currently being evaluated as an anti- biofilm forming agent with initial successes against MRSA biofilm formation.
Yeast and Fungi
Drug resistance is also an issue in for currently available fungicides. Although in its infancy, TolaSure has been effective against C. albicans a dimorphic fungus implicated in infections of the skin, genitals, throat, mouth, and blood. It is also implicated in biofilm infections and Crohn’s disease.